With an era ripe for meticulous exploration in the field of organic chemistry, Professor Max Martin Hansmann and his dedicated research team have unwrapped a groundbreaking reagent that allows for the selective addition of carbon atoms to molecular structures. Published in the prestigious journal *Science*, this advancement signifies a pivotal moment for basic research, showcasing the potential of innovative reagents to redefine molecular synthesis. Conducted under the auspices of the European Research Council’s Starting Grant that was awarded to Hansmann in 2022, the endeavor showcases the synergistic efforts of young researchers contributing to monumental scientific innovations.
Dr. Taichi Koike, serving as a postdoctoral researcher and the principal author of this transformative study, is not only at the forefront of this research but is recognized as an Alexander von Humboldt Research Fellow—a testament to the caliber of work emanating from Hansmann’s lab. As the duo collaborates, they shed light on the intricacies of molecular modification, inviting the scientific community to rethink traditional paradigms and embrace more sophisticated methodologies.
Precision in Molecular Modification
Professor Hansmann’s assertion regarding the elegance of modifying molecules at an atomic level cannot be overstated. The ability to alter molecular structures with such precision opens doors to the synthesis of complex pharmaceuticals through shorter sequences, which could optimize efficacy while reducing resource consumption. This prospect alone promises to enhance the reach of pharmaceutical research and formulating while also potentially leading to more environmentally sustainable practices.
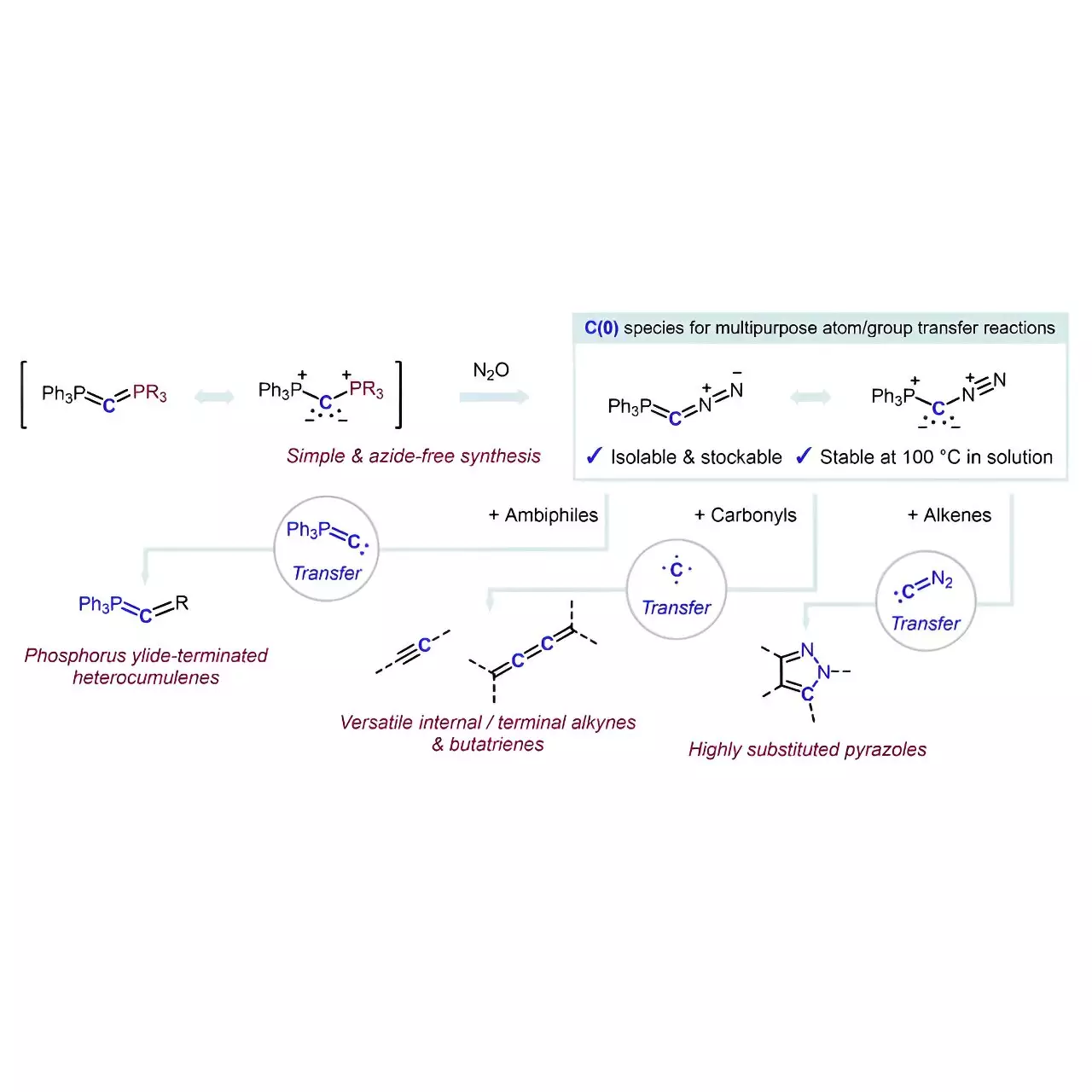
However, the road to the development of reagents capable of selectively inserting carbon atoms into various molecular frameworks has been laden with challenges. It is within this complex landscape that Hansmann’s team made significant strides, successfully synthesizing a reagent that operates as both a source of carbon and a versatile transfer mechanism. The ingenuity of their approach lies in the stabilization of carbon atoms through coordination with neutral electron-donating groups—marking a notable departure from conventional methodologies.
Uncovering the Potential of Carbones
The inventive reagent, characterized by the creation of carbones (L1↔C↔L2), represents an uncharted territory for carbon atom sources. Prior literature has scarcely explored the potential applications of carbones, which speaks volumes about the novelty of this research. By synthesizing a reagent where the carbon atom is flanked by two simple and labile groups, namely phosphine (PPh3) and nitrogen (N2), the researchers not only succeeded in creating a stable platform for carbon transfer but also navigated the safety concerns typically associated with azide reactions present in diazo compound syntheses.
The straightforward synthetic protocol leading to the crystalline and isolable reagent Ph3P=C=N2 exemplifies the Hansmann team’s commitment to elegance and simplicity. Their strategy involved a formal exchange reaction between carbodiphosphorane Ph3P=C=PPh3 and nitrous oxide (N2O), which deftly eliminates the necessity for azides—a major safety hazard in many conventional reactions.
Expanding Horizons in Reactivity
The capacity of this newly developed reagent as a selective transfer tool for carbon fragments opens exhilarating vistas in organic synthesis. The seamless transfer of phosphorus ylide to ambiphiles results in phosphorus ylide-terminated heterocumulenes, while nitrogen transfers to alkenes cleverly synthesize multi-substituted pyrazoles. Notably, the carbon transfer reactions with carbonyl compounds further reveal the reagent’s utility, resulting in a spectrum of alkynes or even the complex butatrienes, which are significantly more challenging to synthesize through traditional routes.
The promise inherent in Hansmann’s findings goes beyond scientific curiosity; it charts a realistic pathway toward the development of complex pharmaceuticals, potentially revolutionizing the industry. With scientists facing increasingly stringent regulations for synthetic processes, the introduction of such a versatile reagent could ease the burden of compliance while encouraging innovative thought amidst the exploration of new drug candidates.
Each of these findings positions Hansmann and his team not only as pioneers in the field but as examples of how targeted research aims to transform the landscape of organic chemistry, inviting peers to keep pace with the rapid evolution of methodologies and applications.