In a compelling advancement for sustainable energy solutions, chemists are making significant strides in utilizing waste materials, specifically carbon dioxide, to produce high-value commodities like methanol. The recent research conducted by a multidisciplinary team of scientists, highlighted in Nature Catalysis, reveals an innovative approach to synthesizing methanol directly from CO2 using electrical stimulation. This is borderline revolutionary for both the scientific community and the energy sector, as it opens new avenues for reducing greenhouse gas emissions while generating useful fuels.
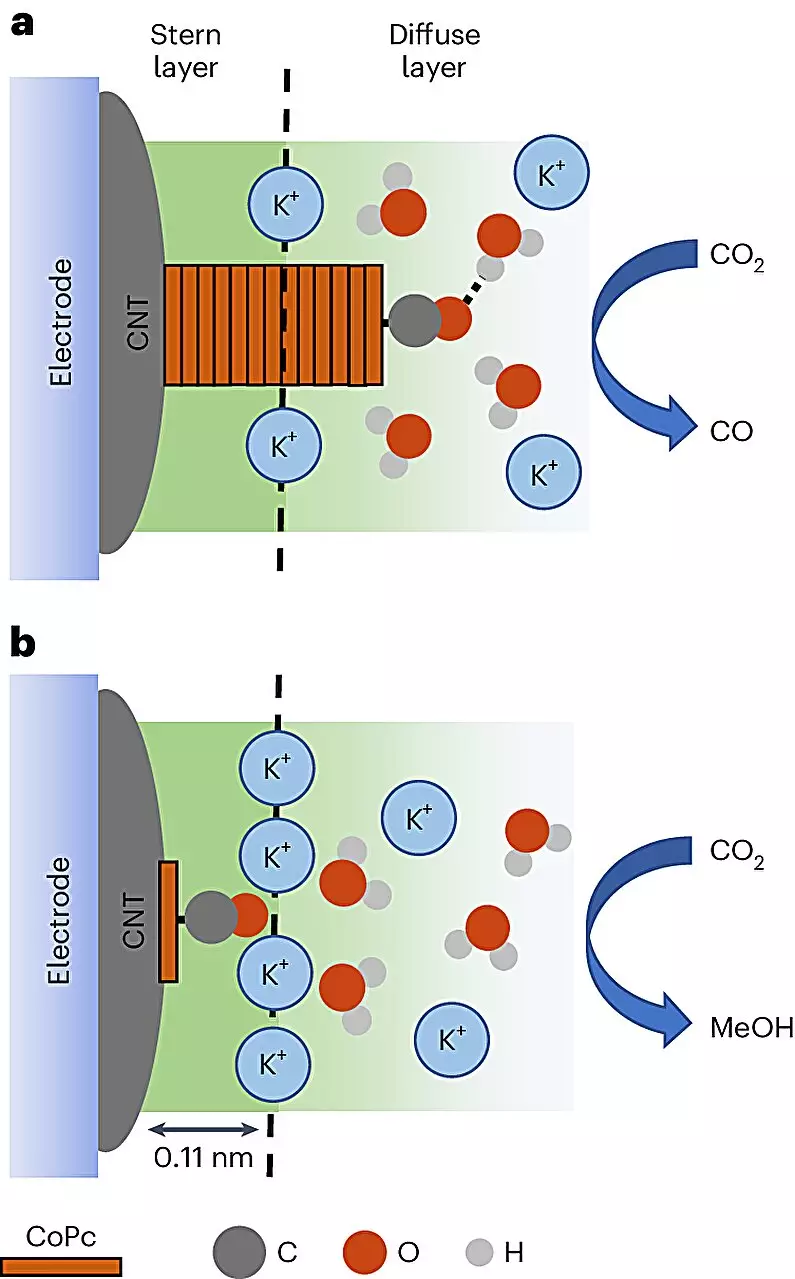
The process hinges on the utilization of cobalt phthalocyanine (CoPc) molecules layered on carbon nanotubes, which display extraordinary electrical properties. By running an electrical current through an electrolyte solution, the CoPc molecules are activated to capture electrons, facilitating the conversion of carbon dioxide into the desired liquid fuel, methanol. The implications of this method go beyond mere experimental success; they may well signal the advent of new techniques for efficient waste-to-fuel transformations.
Understanding Reaction Mechanisms at a Molecular Level
One of the most striking revelations from this study is the researchers’ ability to visualize the chemical reactions in real-time. For the first time, scientists were able to observe how CoPc molecules transitioned into methanol or, undesirably, carbon monoxide. This insight elucidates the significance of the reaction environment and its influence on the outcome, emphasizing the need to refine our current understanding of catalytic processes.
The research employs cutting-edge in-situ spectroscopy techniques, allowing scientists to correlate specific vibrational signatures with the behavior of molecules in distinct reaction environments. This level of analysis equips researchers with the tools necessary to decipher the complex dance of electron transfer and reaction pathways. According to the lead author of the study, Quansong Zhu, this correlation between molecular environment and product specificity holds immense promise for optimizing various chemical transformations.
The Economics of Methanol: Energy Density and Versatility
Methanol is more than just a product of this innovative reaction; it is a powerful alternative fuel with considerable energy density. With the world increasingly shifting towards renewable resources, methanol synthesized from renewable electricity presents an affordable and versatile option for power generation, heating, and even as a fuel for transportation. As Robert Baker, co-author and chemistry professor, elucidates, methanol not only serves as an alternative to conventional fuels but also carries benefits for long-term storage of renewable energy.
Moreover, the potential applications of methanol extend into various realms, including shipping and aviation, where the need for low-cost and effective energy sources is paramount. As researchers delve deeper into understanding the mechanisms that enhance methanol formation, we stand at the precipice of a shift in energy paradigms, suggesting that ecological responsibility and economical productivity can indeed go hand in hand.
Challenges Ahead: Cations and Chemical Interactions
While the findings are promising, researchers have identified that supercharged particles known as cations play a pivotal role in the methanol formation process. These interactions require further exploration to fully harness their benefits and understand their effects on the efficiency of the reaction. Baker notes the challenges inherent in delineating the roles these cations play, hinting at a need for additional studies to unlock their potential. This underscores the complexity of chemical processes and advocates for a holistic approach to research in catalysis.
By strategically controlling the distribution of CoPc on the carbon nanotube surface, researchers can effectively manipulate reaction conditions to maximize methanol production, heightening the profitability of catalysis techniques across other domains. Tuning the environmental factors within the reaction apparatus to favor methanol synthesis presents an opportunity to rethink not only catalysis but also the sustainability of chemical manufacturing as a whole.
Envisioning a Future of Sustainable Chemistry
The implications of this research echo far beyond the laboratory, pointing towards a future where carbon dioxide can be a valuable resource rather than a liability. Beyond simply eliminating a greenhouse gas, the ability to convert CO2 into useful fuels like methanol could drastically reshape the landscape of energy sources. Innovations in this arena may pave the way for sustainable practices that can mitigate climate change while simultaneously addressing energy demands.
Scientists are excited to build upon these findings, pushing the frontiers of chemistry to harness waste materials creatively. With ongoing studies that continue to refine our understanding of catalytic processes, the potential for game-changing technologies has never seemed more promising. As we evolve our approaches to energy and sustainability, this research could indeed represent a critical leap toward a more responsible and innovative future in chemistry and energy production.