Seawater electrolysis presents a viable pathway to decarbonizing global energy systems. This process leverages abundant oceanic resources to generate hydrogen, a cleaner alternative to fossil fuels. However, several obstacles complicate direct seawater electrolysis, such as the susceptibility of anodes to corrosion by chloride ions, unintended oxidation reactions, and the substantial costs associated with high-performance catalysts. Overcoming these challenges is critical to unlocking the full potential of hydrogen production from seawater.
Recent advancements have spotlighted self-supported nickel-iron (NiFe) catalysts as promising candidates for both hydrogen evolution reaction (HER) and oxygen evolution reaction (OER). The intrinsic activity and affordability of these materials have drawn considerable interest from the scientific community. However, the quest for improved stability and performance under seawater conditions continues to motivate research. One innovative solution involves utilizing wood-based carbon (WC) as a substrate for these catalytic materials, leveraging its hierarchical porous structure and impressive electrical conductivity to enhance performance.
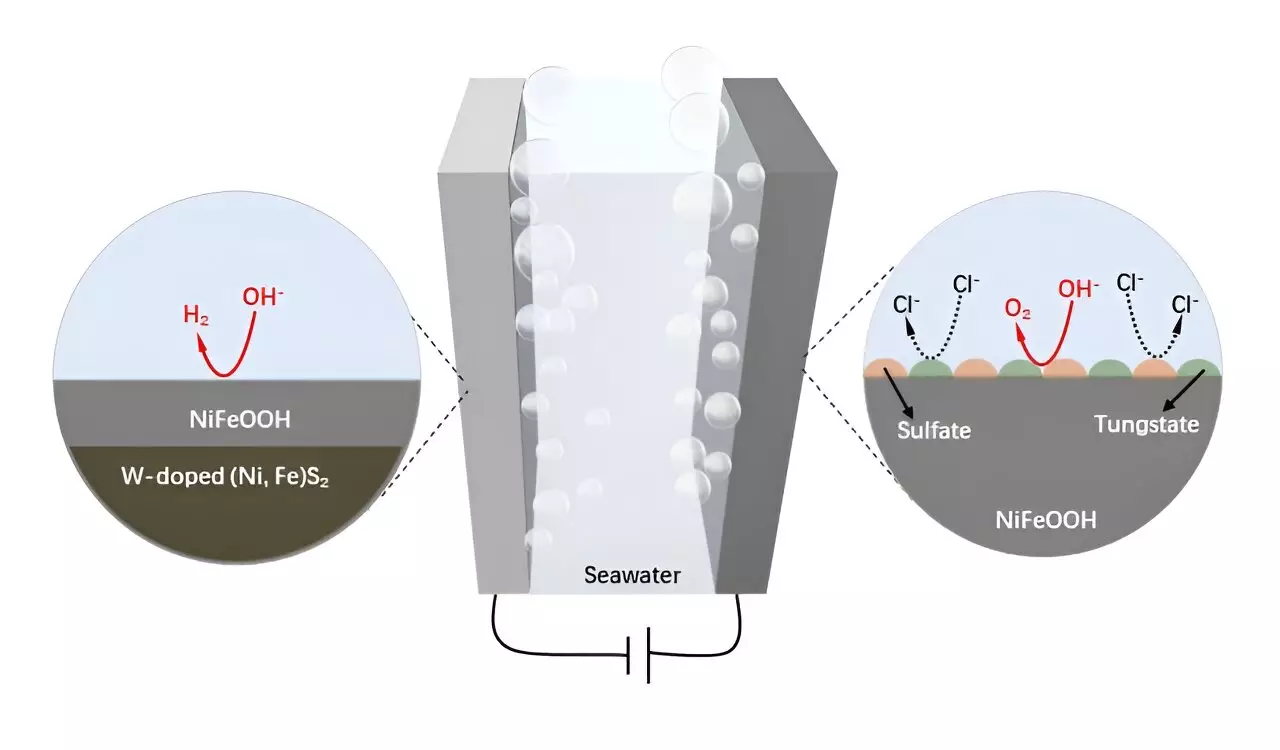
A groundbreaking study led by Professors Hong Chen, Bing-Jie Ni, and Zongping Shao represents a significant progression in this area. Their collaborative effort yielded a W-doped NiFe sulfide catalyst supported by wood-derived carbon (W-NiFeS/WC), which addresses the critical issues of anode stability and durability in harsh seawater conditions. Their findings, released in the prestigious journal *Science Bulletin*, emphasize the transformative impact of tungsten in augmenting the anti-corrosion properties of NiFe catalysts.
The fabrication method for the W-NiFeS/WC electrode involved a meticulous process of impregnation and sulfidation. This production method led to the formation of a robust, three-dimensional hierarchical porous structure characterized by precisely aligned microchannels. These features not only optimize the distribution of W-NiFeS nanoparticles but also significantly improve the overall efficiency and conductivity of the electrode. The resulting configuration supports enhanced electrocatalytic performance, particularly in alkaline seawater environments.
The W-NiFeS/WC electrode exhibits remarkable efficiency and durability in both OER and HER processes, distinguishing itself from traditional catalysts in performance metrics. According to Zhijie Chen, the primary author of the study, the electrode’s inherent structure evolution promotes the formation of protective tungstate and sulfate species on the surface of the active materials, thereby enhancing operational longevity. Furthermore, the low-cost fabrication process combined with superior catalytic performance underscores the electrode’s potential in advancing sustainable hydrogen fuel production.
This research not only enhances our understanding of electrocatalysis but also exemplifies a circular economy approach by repurposing wood waste into innovative and efficient catalytic solutions. By minimizing resource waste and promoting sustainable practices, this work stands at the intersection of ecological responsibility and technological advancement. The implications of these findings extend beyond the realm of academics; they offer a promising pathway toward green hydrogen production that could reshape the future of energy.