The ongoing crisis surrounding plastic waste is both monumental and alarming. Since the inception of mass-produced plastics in the 1950s, an astonishing 8.3 billion metric tons of polymers have been generated. Yet, only a paltry 600 million metric tons of this material have undergone successful recycling. The overwhelming majority has wound up in landfills or has been incinerated, unleashing toxins that pollute our environment and compromise ecosystems. As concern over plastic pollution escalates, the imperative to find efficient recycling methodologies has never been more urgent.
Dr. Junpeng Wang’s Revolutionary Approach
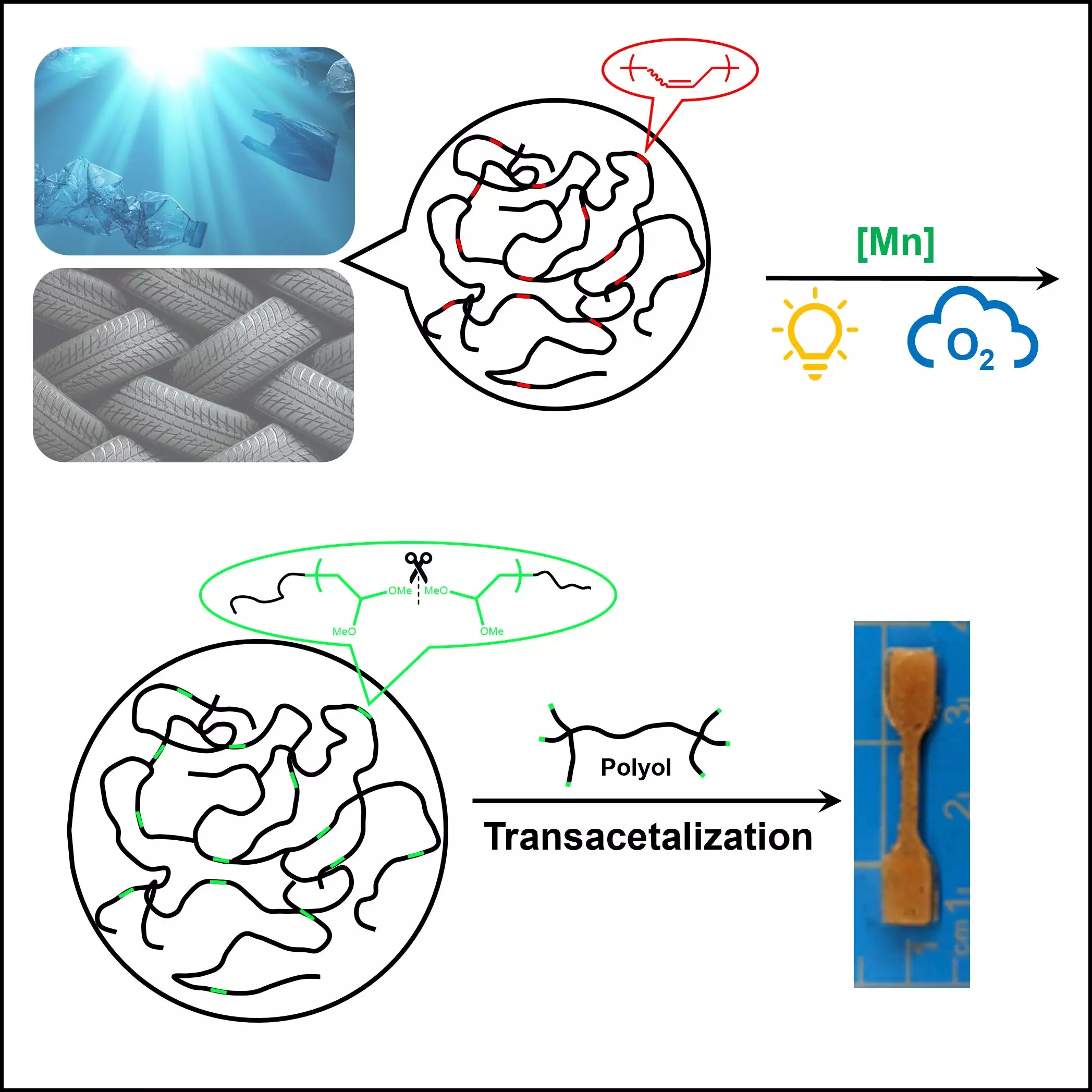
Recent research spearheaded by Dr. Junpeng Wang, an influential figure in polymer science at The University of Akron, has unveiled a fresh perspective on tackling this dilemma. Together with a dedicated team of students and scholars, Dr. Wang presents a promising approach that primarily focuses on unsaturated polymers like rubber and various plastics. Demonstrated in the esteemed journal Cell Reports Physical Science, this research harnesses the power of oxygen and light in a novel process designed to degrade these persistent materials effectively.
Challenges with Existing Recycling Methods
Historically, the recycling of polyolefins—polymers that dominate global production—has presented staggering challenges. Their robust hydrocarbon structures render them stable and, unfortunately, resistant to traditional recycling practices. Conventional techniques such as ozonolysis and permanganate oxidation are fraught with their own set of complications; they often demand harsh, environmentally detrimental conditions and yield unwanted by-products that add insult to injury. Therefore, the need for an eco-friendlier, scalable solution has become paramount.
A Breakthrough in Controlled Polymer Degradation
Dr. Wang’s innovative study breaks new ground by proposing a method that employs a catalyst activated by light to facilitate the breakdown of unsaturated polymers. This system operates effectively at room temperature and circumvents the necessity for extreme pressures or excessive energy inputs, marking a significant stride forward in polymer chemistry. By optimizing oxidation processes to utilize abundant and eco-friendly molecular oxygen, the researchers have underscored the importance of developing sustainable practices in the fight against plastic pollution.
Potential Implications and Future Directions
The implications of this research are far-reaching. If this controlled degradation method can be effectively scaled, it has the potential to revolutionize how we approach plastic recycling. It not only enhances our understanding of polymer degradation but also paints a hopeful picture of a future where less plastic is discarded and more materials are reclaimed and reused. In an era teeming with environmental challenges, the work of Dr. Wang and his colleagues serves as a beacon of innovation, illuminating pathways toward sustainable practices that could drastically alter the lifecycle of plastics.
As advancements in polymer science unfold, we must continue to support and recognize the importance of such groundbreaking research. Innovations like these could very well shape a more sustainable and responsible approach to material consumption in the 21st century, rendering our planet a healthier place for generations to come.