In a bold stride towards combating environmental pollution, a research team from the Ulsan National Institute of Science and Technology (UNIST) has unveiled a groundbreaking catalyst that effectively emulates the functionality of natural enzymes. This innovative catalyst addresses the urgent need for efficient methods to break down harmful hydrocarbons, thereby fostering a more sustainable and energy-efficient approach to pollution reduction.
The work, spearheaded by Professor Jaeheung Cho from UNIST’s Department of Chemistry, has captured significant attention within the scientific community for its potential implications on industrial applications. By principally focusing on the catalytic properties of metalloenzymes—widespread entities in nature that play a crucial role in various biochemical reactions—the research team has manipulated the go-to elements to enhance performance significantly. Their findings were published in the prestigious *Journal of the American Chemical Society* on June 3, 2024, marking a pivotal moment in pollution control technology.
Innovative Mechanisms at Play
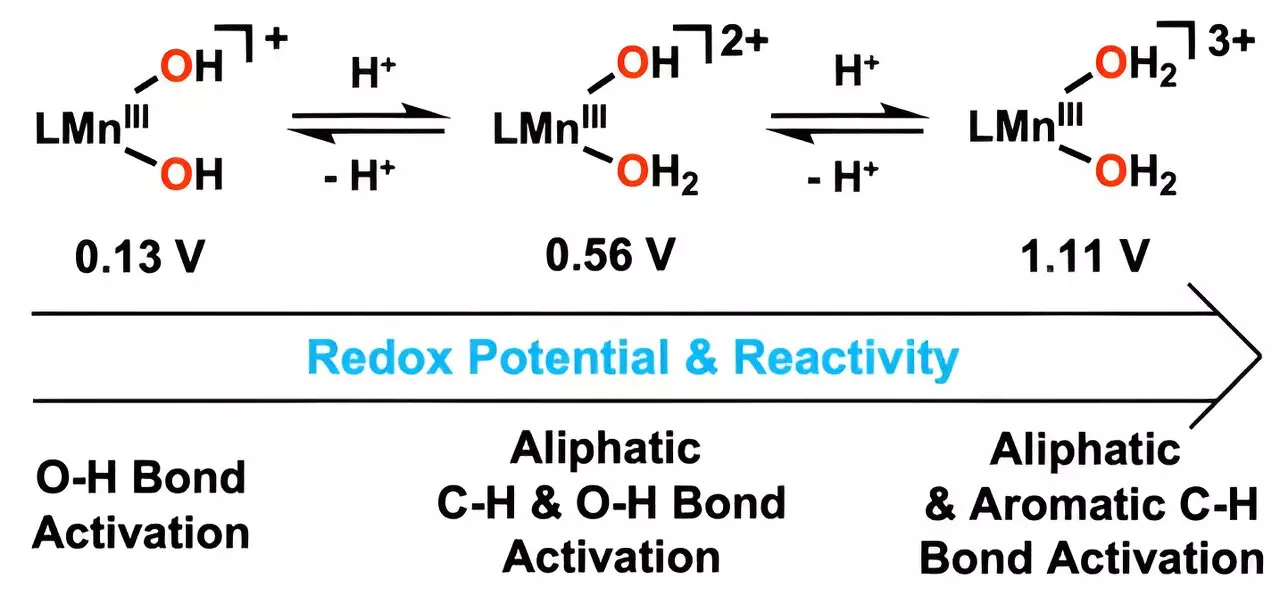
The catalyst’s advanced capabilities stem from the ingenious incorporation of hydrogen ions with hydroxo ligands, leading to the formation of metal-bound water molecules. This innovation allows the catalyst to oxidize carbon–hydrogen (C–H) bonds effectively, achieving this at notably lower temperatures than traditional methods. Such a characteristic could drastically transform existing industrial processes, particularly those involved in hydrocarbon breakdown.
The addition of hydrogen ions has been a key component, increasing the manganese catalyst’s ability to engage with oxygen–hydrogen bonds. Consequently, this enhancement leads to accelerated reaction rates, a transformative feature that can optimize efficiency in chemical production. By modifying the ligands, the research team succeeded in raising the reduction potential of manganese, a crucial factor in the effectiveness of this new catalyst.
Game-Changing Applications and Impacts
One of the standout achievements of this research lies in testing the catalyst on anthracene, a compound renowned for possessing robust carbon–hydrogen bonds. The results indicate successful oxidation at low temperatures, ultimately rendering the otherwise toxic compound non-hazardous. Such accomplishments highlight the catalyst’s ability to decompose not only toxic aromatic hydrocarbons but also those compounds that defy breakdown via conventional methods.
Professor Cho’s assertion that this represents the first occurrence of a manganese(III) complex reacting with aromatic hydrocarbons at lower temperatures underscores the revolutionary status of this research. This catalyst’s advancement also lays the groundwork for the development of industrially viable metal catalysts, promising a future where strong carbon–hydrogen bonds can be dismantled intelligently and sustainably.
While challenges in the realm of environmental chemistry persist, this new catalyst embodies hope for a cleaner planet. The ability to manage and break down noxious compounds without excessive energy loss redefines our expectations for pollution management and provides a promising trajectory for future research in catalytic processes. As more breakthroughs in this area arise, the potential to significantly reduce environmental toxins grows ever more tangible.