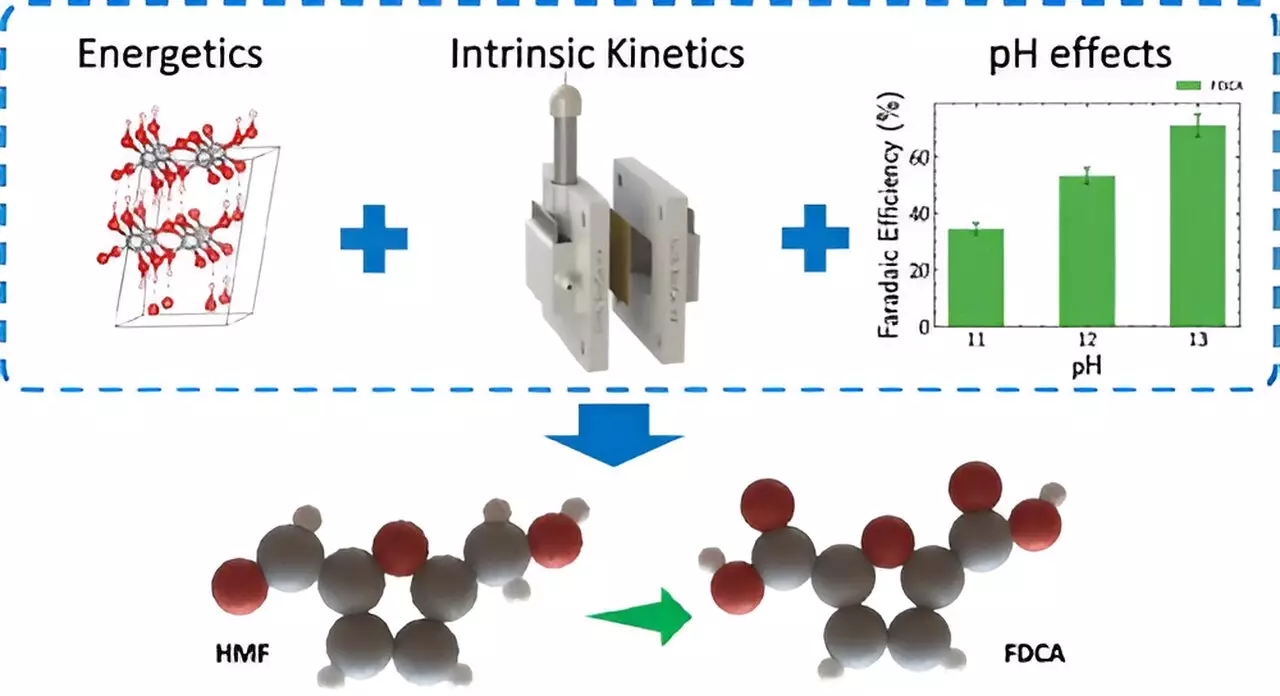
Recent advancements in electrochemical techniques offer a revolutionary shift towards more sustainable and energy-efficient chemical production. Researchers at Lawrence Livermore National Laboratory (LLNL) have pioneered a novel methodology employing thin film nickel anodes, a breakthrough that holds the promise of significantly reducing the environmental impact of chemical synthesis processes. The innovative use of thin films provides a stable and uniform surface for the catalyst, which is essential in enhancing our understanding of the underlying reactions. LLNL postdoctoral researcher Aditya Prajapati, a co-author on the recent publication in ACS Catalysis, emphasized that this approach simplifies experimental conditions, thereby allowing researchers to better assess catalyst performance without the interference of irregular surface textures.
A pivotal issue in conventional electrolyzers—the devices that facilitate the conversion of electrical energy into high-energy molecules—lies in their energy efficiency. Traditionally, these systems face substantial energy losses during the oxygen evolution at the anode. In a compelling development, the LLNL team discovered that substituting the energy-intensive oxygen evolution reaction with biomass oxidation not only improved energy efficiency but also lowered energy consumption by over 50%. This advancement addresses the dual challenges of enhancing performance while ensuring sustainable conversion methods.
At the heart of this innovative process is the transformation of 5-Hydroxymethylfurfural (HMF), a chemical derived from biomass, into 2,5-Furandicarboxylic acid (FDCA). FDCA is a critical building block in the production of sustainable polymers, such as PEF (polyethylene furanoate), which represents a greener alternative to traditional petroleum-based plastics. The implications of this research extend far beyond chemical production; they contribute to mitigating climate change by reducing both carbon emissions and dependency on fossil fuels.
Nitish Govindarajan, another key author of this work, remarked on the significant advantages of the new electrochemical methodology. Unlike conventional processes that often necessitate high temperatures and result in harmful byproducts, the team’s approach stands out for being both cleaner and more efficient. By exploiting biomass as the starting substrate, the method aligns perfectly with the principles of a circular economy, promoting the use of renewable resources while simultaneously curtailing reliance on finite fossil fuel supplies.
One of the most promising aspects of this method is its potential to achieve a zero-carbon footprint, especially when integrated with renewable energy sources. By minimizing energy requirements in chemical production and using renewable feedstocks, this process not only reshapes how chemicals are produced but also has the potential to revolutionize the industry, steering it towards sustainability.
The LLNL study, which also includes contributions from researchers at Université de Montréal and the University of Bonn, showcases the importance of collaborative science in tackling complex challenges. As the global demand for more sustainable chemical processes continues to rise, such research initiatives are critical for paving the way toward environmentally responsible industrial practices. With ongoing developments in electrochemical methods, the future of chemical production looks promising, combining efficiency, sustainability, and innovation in one comprehensive strategy.