The phenomenon of self-organization plays a pivotal role in understanding how life emerges from matter that is, by definition, lifeless. A recent study has revealed a previously unknown mechanism of self-organization integral to bacterial cell division. Conducted by the team led by Professor Anđela Šarić at the Institute of Science and Technology Austria (ISTA), this research underlines a fascinating principle of active matter: specifically, how misaligned protein filaments contribute to the formation of organized structures through a process of “dying to align.” This groundbreaking work holds potential applications in developing synthetic self-healing materials and exploring the broader concepts of life-like systems.
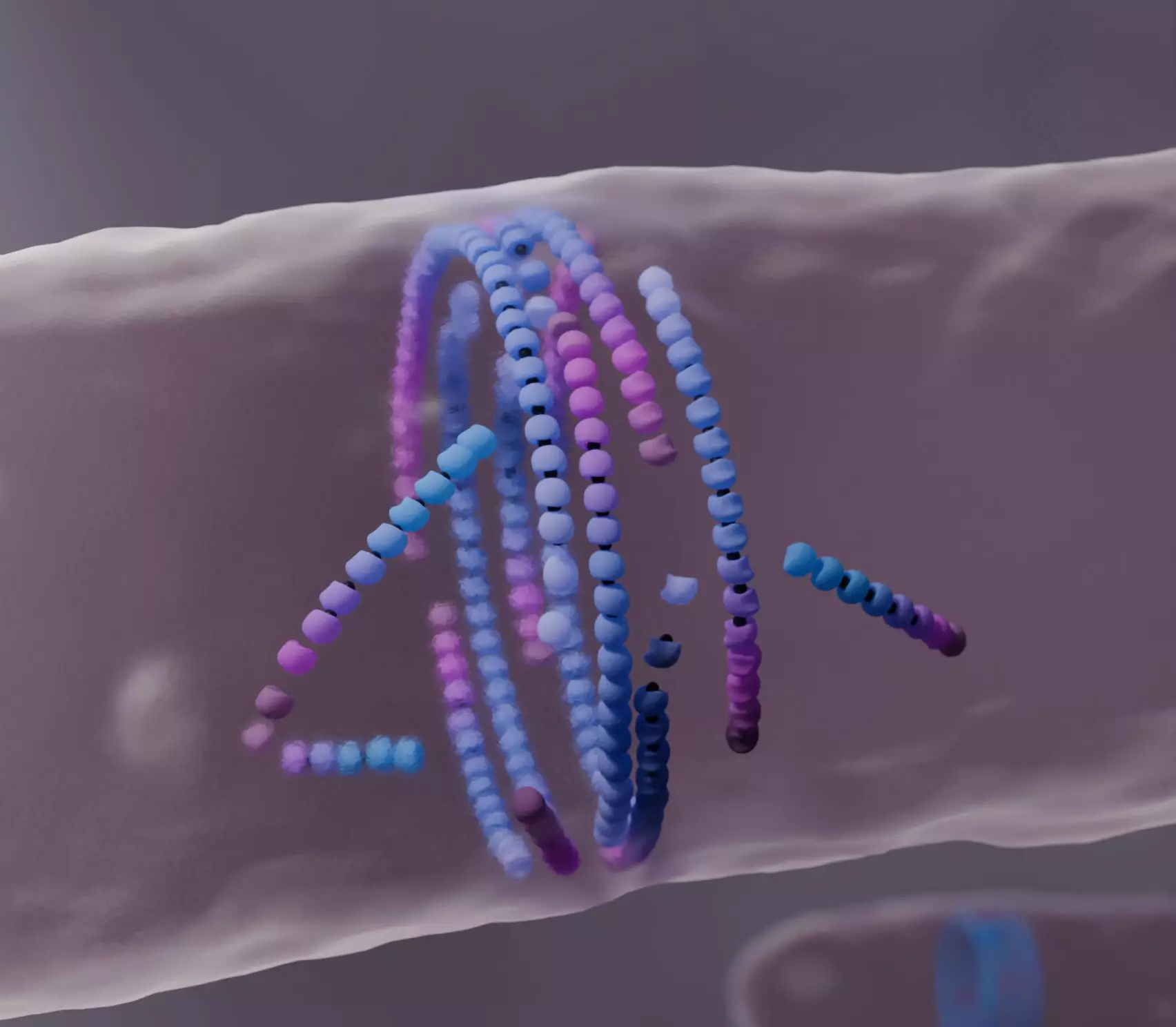
A key player in bacterial division is a protein known as FtsZ, which assembles into a dynamic ring structure at the center of dividing cells. This FtsZ ring acts as a scaffold that guides the formation of a new cell wall, effectively separating the daughter cells. Although researchers have been aware of the important role FtsZ plays, several critical aspects of its self-assembly process were not well understood until now. The Šarić group’s new computational model sheds light on how these filaments behave during cell division, particularly when faced with obstacles.
The concept behind the model focuses on “treadmilling”—a process where subunits are constantly added to one end of a filament while being removed from the other, allowing for dynamic assembly despite overall filament stability. Traditional models have viewed this movement as unidirectional propulsion, overlooking the filaments’ potential to respond to their environment in more complex and biologically relevant ways.
The innovative aspect of this research is the discovery that misaligned FtsZ filaments respond to obstacles by “dying.” This death is not permanent; instead, it leads to a reorganization and alignment that enhances the overall structure of the FtsZ ring. The researchers found that when a misaligned filament encounters an obstacle, it dissolves rather than pushing against it, allowing better-aligned filaments to take precedence. This phenomenon raises intriguing questions about the resilience and adaptive capabilities of biological systems, wherein death contributes to the health of the group.
This self-organizational behavior is akin to a natural selection process at the molecular level, where misaligned components are eliminated, making way for a more robust assembly. The implications of such dynamics suggest a higher level of sophistication in how active matter operates, revealing a relationship between individual and collective behavior that has not been fully appreciated in prior models.
What stands out in this investigation is the strong collaborative spirit fostered between different research groups to validate the computational findings. The Šarić group collaborated with experimental teams from the University of Warwick and ISTA, led by Seamus Holden and Martin Loose, respectively. This partnership proved invaluable as it provided experimental evidence that mirrored the computational results, confirming the model’s predictions.
Conference interactions allowed the researchers to establish connections across disciplines, which was crucial for validating their theoretical framework. The integration of insights from molecular biology, physics, and computational modeling exemplifies the need for interdisciplinary approaches in tackling complex biological questions.
The implications of this research extend beyond understanding bacterial biology. The findings suggest possible pathways for constructing synthetic materials that mimic biological processes—particularly self-healing materials capable of addressing damage through self-regeneration. By investing energy in the turnover of materials rather than their movement, researchers can tap into new frameworks to create living-like systems from non-living components.
Professor Šarić and her team aim to delve deeper into how the FtsZ ring contributes to building the new cell wall. Understanding this will not only provide further insights into bacterial cell division but may also stimulate advances in synthetic biology, leading to innovations in material science and regenerative medicine.
The exploration of active matter through the lens of bacterial cell division has yielded rich findings regarding self-organization. By revealing the “dying to align” mechanism of FtsZ filaments, researchers are unearthing fundamental principles that govern life at a cellular level. This work emphasizes the intricate balance between individual components and collective organization in biological systems and sets the stage for future innovations in synthetic systems that echo the complexities of life. With collaborative efforts bridging various fields of research, the future holds immense potential for discovering and harnessing the secrets of self-organization in both biological and synthetic contexts.