In a remarkable study published in *ACS Catalysis*, Professor Yan Wensheng and his research team from the University of Science and Technology of China (USTC) have made significant strides in understanding single-atom catalysts, focusing specifically on the enigmatic relationship between metal loading and acidic oxygen evolution reaction (OER) activity. Traditional challenges in creating highly effective catalysts have kept researchers at a standstill, particularly when it comes to maintaining stability at various metal loadings. This study employs innovative approaches to illuminate the underlying principles that dictate how metal concentration impacts catalytic performance.
The Significance of Metal Loadings
Metal loadings are critical when it comes to enhancing the efficacy of catalysts. The research embraced a P-anchoring strategy, leading to the synthesis of a suite of Iridium (Ir) single-atom catalysts, notably labeled as x-IrPN3@C, featuring metal loadings ranging from 5% to an impressive 21%. This presents a paradigm shift in how catalysis is approached, particularly with respect to single-atom systems where the precision of metal atom distribution is a foundational aspect. The study addresses the delicate balance: while it may seem intuitive that an increase in metal concentration would correspond positively with catalytic activity, real-world chemistry tells a nuanced and intricate story.
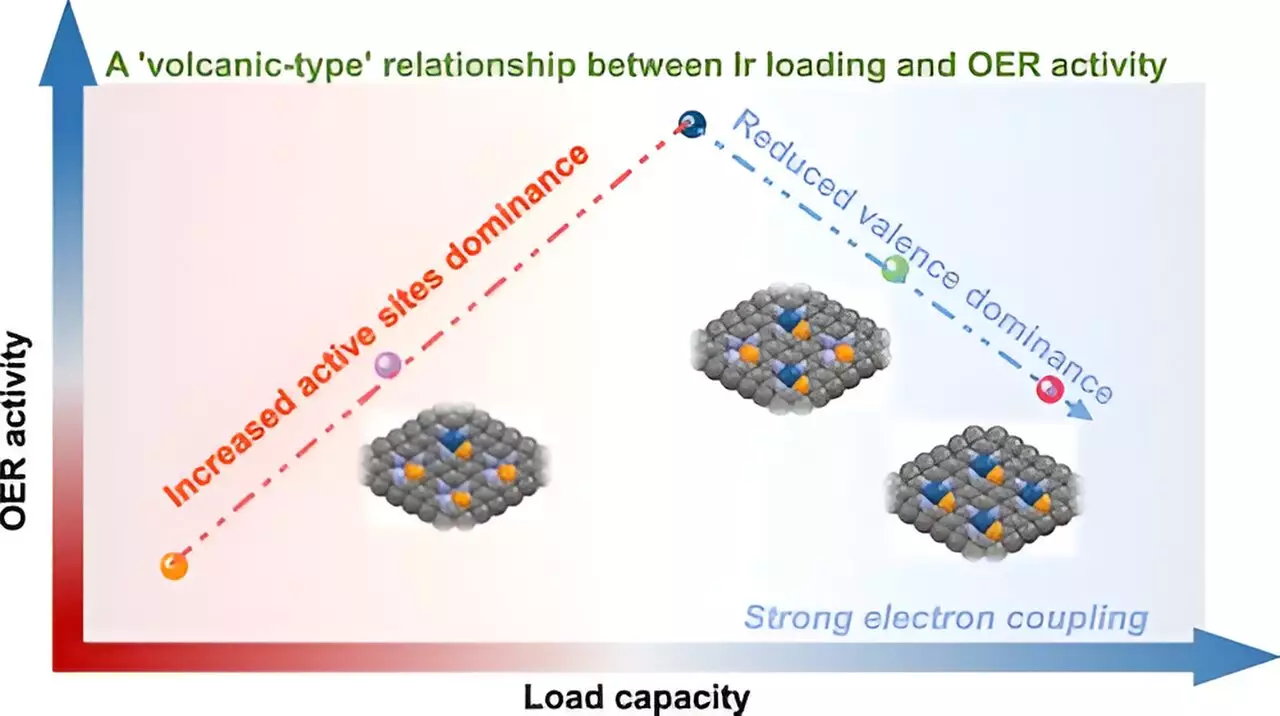
The Volcano-Type Relationship
The most astonishing revelation is the “volcano-type” relationship unveiled between metal loading and catalytic activity concerning the OER. This term, prevalent in catalytic studies, signifies a bell-shaped curve where activity increases with metal load until a peak is reached, after which further increases lead to a decline in performance. What sets this research apart is the identification of key electronic interactions among the metal atoms at higher loadings, central to understanding their macroscopic behavior. The team’s use of advanced analytical techniques like synchrotron radiation XAS spectroscopy allowed them to observe that the Ir-P coordination structure plays a pivotal role in maintaining stability and preventing aggregation of atoms at elevated loadings.
Mechanistic Insights and Implications
Delving deeper, the researchers noted that when the Ir loading crosses a critical threshold, adjacent atoms start to interact more intensely. This results in diminished valence state for Ir, which directly correlates to a decline in OER activity. Such insights carry significant implications for catalytic design, steering scientists away from simply maximizing metal content. Instead, they highlight the necessity for a balanced approach to ensure optimal performance while avoiding pitfalls associated with excessive loading.
In the grand scheme of catalysis, this research serves as a beacon for future innovations. By providing a theoretical framework based on empirical evidence, the findings can aid in the engineering of more effective and cost-efficient catalysts that can be utilized in a variety of chemical reactions. Recognizing the intricate dance of atomic interactions reinforces the understanding that in catalysis, more isn’t always better; instead, a methodical approach to optimize specific conditions is essential for breakthrough advancements in the field.